ADME and Rational Chemical Design
Introduction ADME characterizes drug and chemical uptake into a body and, therefore, needs to be considered before designing and creating new chemicals, or re-classifying use parameters for existing chemicals.
Learning Outcomes:
- Investigate how ADME properties should influence chemical design and creation.
- Associate different physicochemical properties with absorption.
Background Information: ADME stands for Absorption, Distribution, Metabolism, and Excretion. ADME is a term especially known in pharmacology and drug design, but that also has significant applications in both toxicology and green chemistry design. ADME “describes” the effectiveness of the drug by characterizing its toxicokinetics (uptake and fate of a chemical in the body). So before developing any chemical or drug, chemists should consider several metrics:
- the rate of the uptake of the molecule
- how fast and where it is distributed
- how fast is it metabolized
- how fast can it be excreted from the body.
In the realm of pharmacology, knowing these rates helps medicinal chemists design drugs which not only are most effective, but more importantly, that target specific tissues. One of the classic examples of how understanding ADME led to an improved chemical design are allergy drugs. Allergy relief drugs, ‘aka’ antihistamines, are widely prescribed for seasonal and perennial allergies. The drug enters the brain, where it prevents histamine from binding to cells and blocks the allergy reaction mechanism. The first generation antihistamine drugs, however, were identified with a side effect. Because of their structure, antihistamines were very likely to accumulate in the brain and affect other receptors, which in turn lead to drowsiness. Understanding ADME allowed scientists to redesign the drug, which now enters the brain, reacts with the appropriate receptors on the cells, and is excreted before it affects other receptors.
Chemists and molecular designers have also used ADME to create guidelines for designing safer industrial chemicals. In this case, chemicals are designed such that they have a minimal effect on humans and environment. Ideally, chemicals shouldn’t bioabsorb (enter the body) in the first place. However if they do, molecular designers want to reduce distribution, reduce bioactivation, and accelerate excretion. Many of the guidelines for chemists are derived from existing physicochemical properties and toxicity data. Examples of some guidelines are summarized in figures 1 & 2.

Figure 1. Desired molecular weight for decreased oral, respiratory, and dermal absorption. Molecular weight is given in Daltons.

Figure 2. Desired solubility for decreased oral and dermal absorption. Solubility is defined through log Pow and measures relative solubility of a chemical in water (a polar molecule) and octanol (a non-polar molecule). For details, refer to Aqueous and Lipid Solubility Module.
By considering physicochemical properties during the design phase of drug or chemical needs, we can reduce negative toxicological impacts and unintended side effects before the product makes it to market and living systems are exposed to a potentially dangerous chemical.
Video: Dr. Jakub Kostal, Associate Research Scientist, The George Washington Unversity and CSO of Sustainability A to Z
Assignment:
- Find a successful example on how understanding ADME led to a better chemical design. Summarize the issue and outcome in 1-2 paragraphs.
- Propose a new chemical (and, for fun, name the chemical!) that will be minimally toxic to humans based on its physicochemical properties. Detail out what properties you chose for this chemical to possess and how that has an impact on its toxicity using ADME properties to guide your decisions.
- There are several parameters which influence ADME. Changing these parameters from not preferred to desired can potentially impact bioavailabiliy and absorption in organism. While molecular weight and solubility significantly contribute to increase or decrease in dermal, respiratory and digestive absorption, below you will find a list of parameters which also need to be considered while designing a molecule.
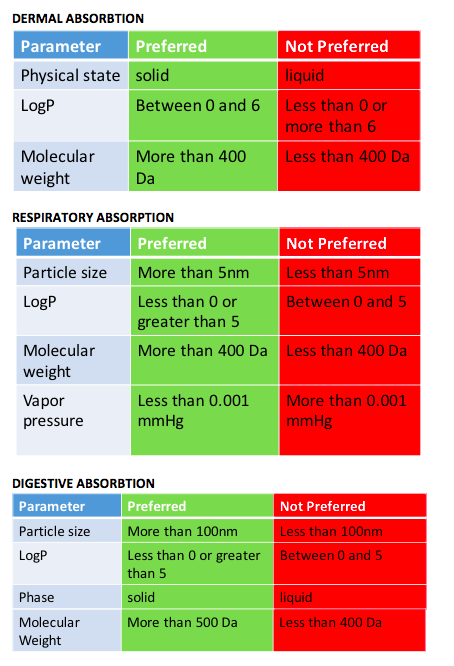
Figure 3. List of other properties which influence ADME along with their preferred and not preferred ranges.
Go to chemspider.com and put in the following CAS numbers (a unique number which is assigned to each chemical):
- 71-43-2 [Benzene]
- 67-68-5 [DMSO]
- 58-08-2 [caffeine]
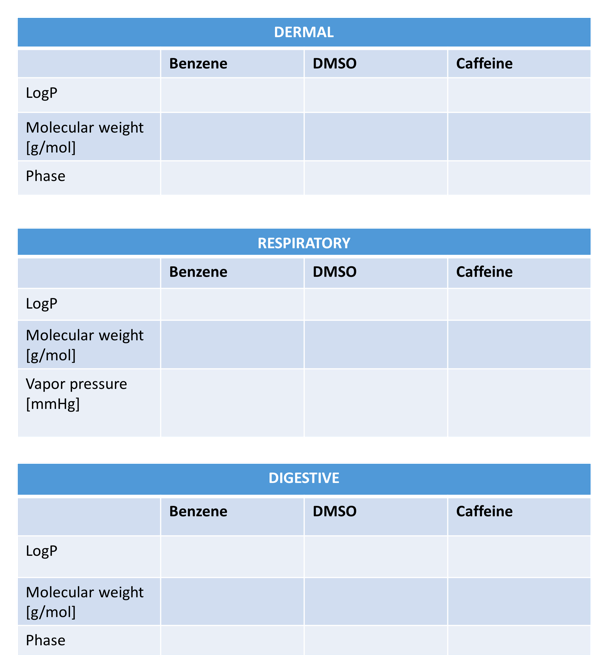
Using discussed parameters and their preferable and non preferable ranges, fill out the following tables to predict respiratory, dermal and digestive absorption of compounds.
a) write down the numbers from chemspider in the appropriate box
b) using the chart above decide whether the property falls in the preferred (green) or not preferred (red) range.
ADME and Rational Chemical Design Answer Key Found HERE
Click here to evaluate this module.
Resources:
- Mulvihill, M., & Vulpe, C. (n.d.). Controlling ADME through chemical design. Retrieved from http://bcgc.berkeley.edu/sites/default/files/Ch298010-2012%20Day%205.pdf
- Di, L. & Kerns, E. H. (2010). Drug-like properties: Concepts, structure design and methods: From ADME to toxicity optimization. eBook. Retrieved from http://www.acad.bg/ebook/cheminformatics/Drug-like%20Properties%20Concepts,%20Structure%20Design%20and%20Methods%20from%20ADME%20to%20Toxicity%20Optimization.pdf
This material is based upon work supported by the NSF Division of Chemistry and the Environmental Protection Agency under Grant No. 1339637.